Chemical Dominoes Board game
During this project we worked in groups of four in order complete a chemistry based board game. In this game you had to include at least 4 different types of reactions including a single replacement, double replacement, production of gas and lighting an LED. The game was also required to be, well, a game where people could play and enjoy playing it. Because of this we chose a candy land style game with an under the sea theme with various under water fish and object representing the different reactions, such as a puffer fish representing the production of gas as it expands with CO2. To move you drew a card from the deck and then preformed the task labeled on it, an example of a move may be go forward 3 spaces or move forward to one 5 tile types such as sand dollar or sea horse. If you landed on a tile after your move that was marked with red x's you would be forced to move back one space and then trigger the nearest reaction. There were also shortcuts on the board that were trigger when you drew a move forward a few spaces card and were on the space that was adjacent to the shortcut. The winner of the game was who ever reached the treasure at the end of the board.
Concepts
Atoms: The building block of basic matter. Everything you can see is made of atoms. Atoms are composed of protons, neutrons and electrons
Protons: Part of an atom, is one of the 2 subatomic particles that make up the nucleus of the atom. It has a positive charge and is the attribute that defines an element
Neutrons: Part of an atom, is one of the 2 subatomic particles that make up the nucleus of the atom. It has a neutral charge and when changed a different isotope is created.
Electrons: Part of an atom, is the subatomic particle that is out side of the nucleus makes up the majority of the volume in an atom. It has a negative charge and when changed it makes the element become a new ion.
Element: Any piece of matter composed of only one type of atom. An example of this would be something like pure nitrogen gas which contains only nitrogen atoms. You cannot divide a element into smaller different parts through chemical change.
Compound: A molecule composed of multiple types of elements, meaning it is composed of more than one type of atom. an example would be hydroxide which contains 1 hydrogen atom and one 1 oxygen atom.
Ions: A form of an element or coumpount with a unique charge. Important to note that all atoms are in a form of an ion and that in order to change a atoms charge one must add or remove electrons as changing a proton will change the element.
Cation: An element or compound with a Positive charge. Most elements that are composed of metals have a positive charge.
Anion: An element or compound with a negative charge. Most elements and compounds which are composed of non-metal are anions but not all.
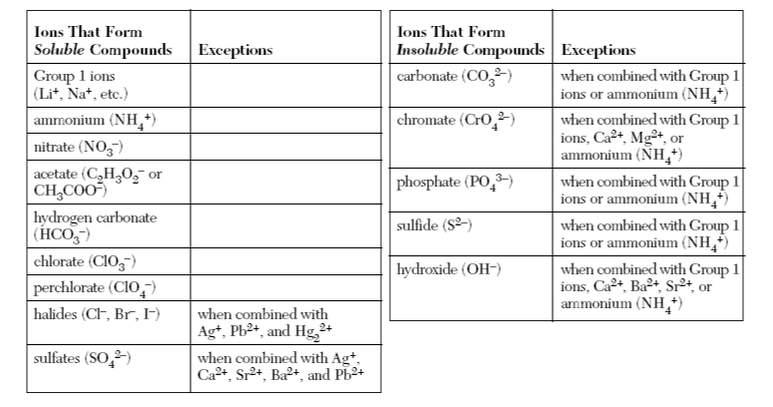
Double Replacement Reaction: A double replacement reaction is reaction that swaps either the cations or anions of a set of aqueous solutions. Reaction occurs if one of the newly formed compounds is insoluble, according to the guidelines below
Protons: Part of an atom, is one of the 2 subatomic particles that make up the nucleus of the atom. It has a positive charge and is the attribute that defines an element
Neutrons: Part of an atom, is one of the 2 subatomic particles that make up the nucleus of the atom. It has a neutral charge and when changed a different isotope is created.
Electrons: Part of an atom, is the subatomic particle that is out side of the nucleus makes up the majority of the volume in an atom. It has a negative charge and when changed it makes the element become a new ion.
Element: Any piece of matter composed of only one type of atom. An example of this would be something like pure nitrogen gas which contains only nitrogen atoms. You cannot divide a element into smaller different parts through chemical change.
Compound: A molecule composed of multiple types of elements, meaning it is composed of more than one type of atom. an example would be hydroxide which contains 1 hydrogen atom and one 1 oxygen atom.
Ions: A form of an element or coumpount with a unique charge. Important to note that all atoms are in a form of an ion and that in order to change a atoms charge one must add or remove electrons as changing a proton will change the element.
Cation: An element or compound with a Positive charge. Most elements that are composed of metals have a positive charge.
Anion: An element or compound with a negative charge. Most elements and compounds which are composed of non-metal are anions but not all.
Double Replacement Reaction: A double replacement reaction is reaction that swaps either the cations or anions of a set of aqueous solutions. Reaction occurs if one of the newly formed compounds is insoluble, according to the guidelines below
Single Replacement: a reaction in which an element replaces an ion of a compound. It takes the form of X+YZ=Y+XZ or X+YZ=Z+YX depending on which type of ion was replaced.
Synthesis Reaction: a reaction in which two or more elements or compounds combine to create a single compound. You can tell if a reaction is a single replacement if it has only a single product.
Decomposition Reaction: a reaction in which a single reactant breaks apart into two or more elements or compounds. It is kind of like the inverse of a synthesis reaction where a synthesis is many smaller parts making a single big thing a decomposition reaction is a single large reactant breaking into many smaller ones. Decomposition reactants are generally exothermic.
Synthesis Reaction: a reaction in which two or more elements or compounds combine to create a single compound. You can tell if a reaction is a single replacement if it has only a single product.
Decomposition Reaction: a reaction in which a single reactant breaks apart into two or more elements or compounds. It is kind of like the inverse of a synthesis reaction where a synthesis is many smaller parts making a single big thing a decomposition reaction is a single large reactant breaking into many smaller ones. Decomposition reactants are generally exothermic.
reflection
This project was a very good project to introduce people into chemistry as it combined a good part of creative thought and chemical science in order to create a good project and over all I feel that I did a good job. For example I always tried to stay active in the project such as fetching items need to preform certain tasks as well trying to reduce the busy work of a single task like cutting things out. I also did a good job at knowing the material for presentations so as to fill in any gaps of information someone may have and help to clarify exactly what each reaction composed. I also struggled in some areas as well however, for example I don't feel I did as good as job as a could when it came to the process of coming up with ideas instead the majority of the creative choices were made by my teammates. Another thing I struggled at is leading as throughout much of the project I was asking others what I should do and how I should do it. For both of these issues I wish to improve on by both being more assertive in leading a group, as well as engaging more with future projects to help stimulate the development of new and creative ideas. Over all I feel this was a very successful project by teaching us about many different reactions but also in engaging us by forcing us to apply it in unique situations.